|
12/2/2023 0 Comments First order half life equation
Rearrangement yields the following: d / = - k dt The decrease in the concentration of A over time can be written as: - d / dt = k Let us use the following chemical equation: A -> products. 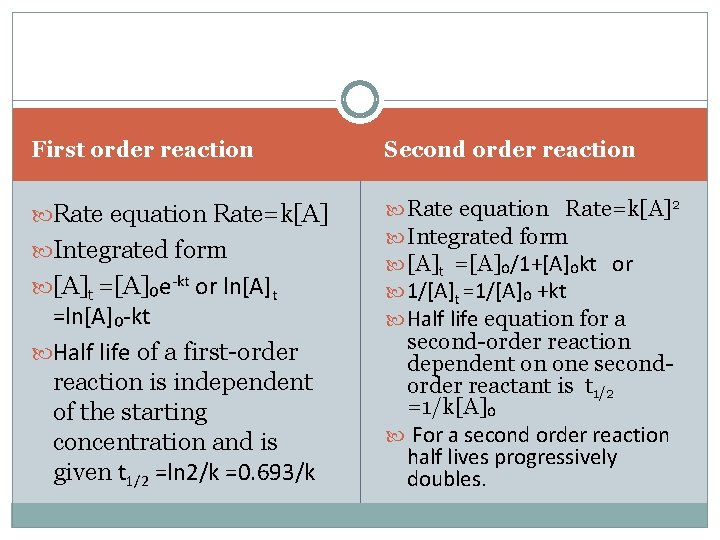 
The Integrated Form of a First-Order Kinetics Equation
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
